South Africa’s first batch of the once-every-six-months HIV prevention shot, lenacapavir (LEN) — paid for with the country’s grant from the Global Fund to Fight Aids, TB and Malaria — will arrive soon, the health department says, although it’s slightly delayed.
Government roll-out is set to start in government clinics within the next couple of months, depending on when the first batch of medicine arrives and by when samples of the first batch have been quality-tested to ensure safe and effective medicine will be distributed.
Results published in 2024 showed that the twice-a-year injection is almost perfect in stopping someone from getting infected with HIV through sex, leading to it being hailed as “the closest thing to a vaccine”. (LEN is not a vaccine; rather, it’s preventive medicine, which works for only as long as someone takes it).
The LEN, sponsored by the Global Fund and enough to phase in about 456 000 HIV-negative people on the jab over two years, will be the branded version bought from the medicine’s original maker, Gilead Sciences. But the shots will be sufficient to cover 3% of the people who need to be put on it between now and 2043 to end Aids as a public health threat by that year, says Lise Jamieson, a modelling scientist at Wits University Health Economics and Epidemiology Research Office.
Ending Aids means new HIV infections, which were around 180 000 in 2024, would have to drop to 65 000 a year, Jamieson has calculated,to bring down the rate of new infections, called the incidence rate, from the current 0.32% to 0.1%.
From next year, the health department will probably start buying generics to add to the branded Global Fund donations and pay for them from its own pocket, with a price tag of around $40 a year (about R640 at the current exchange rate). Exactly when generics will be bought depends on when the manufacturers will have them ready and by when the products will be registered in South Africa.
In a recent development, the South African National Aids Council, in collaboration with four government departments, also announced that they were working with Gilead and local pharmaceutical companies to obtain generic licences to make LEN locally. LEN made in South Africa could be available as soon as next year, which will help companies that do get licences from Gilead to master manufacturing processes, says Unitaid.
In the aftermath of defunding shocks after the US administration change in 2025, “this year will have to be one of strategic rebuilding”, Mitchell Warren, the executive director of the international health organisation, Avac, told Bhekisisa in January.
Countries will have to make smart decisions, he says. And LEN could be that opportunity.
Forecasts from a 2024 study revealed that the jab could prevent between 18% and 33% of new infections, depending on how widely they are used, in three sub-Saharan countries, including South Africa. Modelled data presented at a roundtable event in October, where experts discussed how the country could best use the donated doses it would have available over the next two years, showed similar results.
But to make sure the country doesn’t “miss this moment”, says Warren, the government would have to dig deep into its pocketbook for enough extra doses and campaigns that convince people to use it over the coming years to drive LEN’s roll-out.
Exactly how deep would the health department have to dig though and for how much bang? We take a look at what health economists’ modelling predicts.
How low can the numbers go?
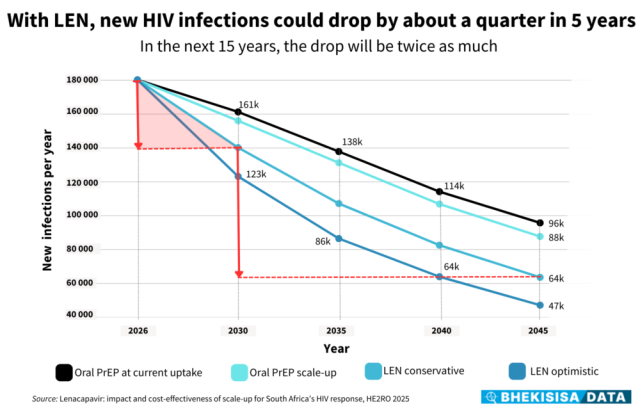
With LEN as part of the HIV prevention basket, new infections could drop by about a quarter in the next five years if our roll-out focuses on people who have a bigger chance of getting HIV, such as teenage girls and young women, gay and bisexual men, female sex workers and pregnant and breastfeeding women, projections show. For this, we’d have to give the shot to, on average, 1.7 million people a year.

If we continue to give LEN to the same number of people, there could be only 64 000 new infections a year by 2045. If, however, 2.9 million people were to use LEN each year — a scenario the modellers called optimistic roll-out — infections could drop to as few as 47 000 by 2045, down three-quarters from the roughly 180 000 where the number sits today.
The case for having LEN in the public system becomes even stronger when compared with other scenarios offering PrEP (pre-exposure prophylaxis, the medical term for using medicine to prevent someone from getting a disease).
In the case of HIV, PrEP medicine prevents someone from getting infected with HIV, the virus that causes Aids. Different types of PrEP registered in South Africa include a daily pill (also called oral PrEP), a two-monthly shot and LEN, which is a six-monthly injection.
South Africa started to roll out the daily HIV prevention pill in 2016; the two-monthly jab hasn’t been rolled out beyond research projects in either the public or private sector.
If we continued to roll out the daily pill in the way we do among the target groups included in the model, there would probably be half as many new infections by 2045 compared with today. A similar drop is seen over this period even if twice as many people started using oral PrEP.
One of the challenges with the pill has been that many people struggle with remembering to take it every day; the less often it’s taken, the less well it works. LEN studies have shown that protection levels with this six-monthly injection are higher than with the daily pill, mainly because it’s easier to adhere to because it has to be taken only twice a year.
The researchers also looked at how rolling out LEN would compare with other strategies to get new infections down.
Results showed that if 95% of people with HIV were on treatment, about 1.4 million new infections could be prevented over 20 years, which is about 2.5 times more than with conservative LEN uptake. When people with HIV use their treatment correctly, the antiretroviral drugs they take stop them from transmitting the virus to other people through sex, because the medicine reduces the amount of HIV in their bodies to extremely low levels.
However, given that the share of people with HIV on treatment has been hovering around 80% in South Africa for some years and the pool growing only a little each year, adding LEN into the mix could help to push down infection numbers much faster. In fact, with LEN, the country’s HIV incidence (the number of new infections in a set period) could drop to below 0.1% by 2039, which would be low enough to mean that Aids would essentially stop being a public health threat.
How much bang can we buy with our bucks?
Getting about the same number of people on the daily prevention pill a year, as in the conservative LEN scenario which would be more than double the number from the targeted groups who use the pill and covering 29% of pregnant and breastfeeding women, it would cost the government about R22 billion extra over the next 20 years and prevent about 120 000 new infections. For roughly the same amount, about four times as many infections could be stopped with LEN.

Spending about three times as much — R64.3bn versus R21.7bn over 20 years — in order to give LEN to roughly twice as many people for twice as long (the optimistic scenario) — 2.9 million people compared with 1.7 million — would prevent about 840 000 new infections over 20 years. This is seven times more than with the conservative uptake.
For comparison, the model also included the two-monthly injection, long-acting cabotegravir (CAB-LA), (as opposed to LEN, which is taken every six months. But investing in CAB-LA would not be a smart move for the health department, results showed: over 20 years it would cost about four times as much as scaling up the daily HIV prevention pill yet prevent only three times as many infections.
At least two Indian drugmakers will make LEN available at $40 a year; the same price as the daily HIV prevention pill. The CAB-LA price used in the model was $180 a year (although the real-life price has since decreased to $160 a year).
The cost to stop one infection is a useful measure to help decision-makers think about spending the budget for HIV prevention. Over 20 years, it would cost around R49 000 to prevent one infection with conservative LEN uptake (if 1.7 million people used LEN each year).
If close to double the number of people were to get LEN in this time, stopping one infection would cost R77 000. The amount includes the cost not only of the medicine but also of campaigns to create demand for it, HIV testing, the salaries of nurses who administer it and so on.
This means both LEN options would give a better return than scaling up the daily HIV prevention pill. But if only a similar amount of money is available as for scaling up oral PrEP, going for a conservative LEN uptake might be the better route for the health department. At around R234 000 to stop one infection — more than four times more expensive than with LEN at low uptakes — CAB-LA would probably not even be considered.
Says Warren about his hope for the coming year: “When history is written, we’ll look back to 2026 and say: ‘South Africa did the right thing in its Aids response and it helped the world when Pepfar wouldn’t.’”
Additional reporting by Jacques Verryn.

This story was produced by the Bhekisisa Centre for Health Journalism. Sign up for the newsletter.
The HIV prevention shot, lenacapavir, will be rolled out at South African clinics within the next couple of months and from 2027, the health department will also buy generics. But how best to spend the HIV prevention budget so that the country can drive infections down as fast as possible? We take a look at what the modelling data shows


![[AFRICA-TICAD] Co-create Innovative Solutions with Africa](https://www.velvetclassic.net/wp-content/uploads/2026/03/YouTube-E88BB1E8AA9EE5AD97E5B995E78988-E88BB1E4BB8FE38391E383B3E38395E68EB2E8BC89-QRE382B3E383BCE38389_20260311-NT1VnV-238x178.png)


